Growth mechanism and active site probing of Fe3C@N-doped carbon nanotubes/C catalysts: guidance for building highly efficient oxygen reduction electrocatalysts†
Abstract
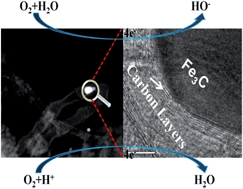
Non-platinum (NP) electrocatalysts with high activity and durability for oxygen reduction reactions (ORR) are required for fuel cells and other renewable systems. To avoid trial-and-error methods and achieve the rational design and synthesis of efficient NP catalysts, in-depth knowledge of the formation/growth mechanism of nanocatalysts and the origin of active sites is highly desirable. Here, we report a new class of NP catalysts with a novel structure of Fe3C encapsulated in N-doped carbon nanotubes/C. We study the formation mechanism of the nanostructure to pave the way for controlled fabrication of high-performance NP catalysts. The encapsulation of iron into carbon occurs during the first step of CNT growth and the surface functional groups on carbon black are identified as being essential for forming CNTs. The catalyst shows ultrahigh catalytic performance in both acid and alkaline media. We also examine the structure–performance dependency. The catalytic performance is highly dependent on the nanostructure and the encapsulation of Fe3C. Fe affects the catalytic performance through electronic effects rather than by directly participating in the active sites. This result is confirmed by DFT calculations, which show an increase in the density of states and a reduction in the local work function, XPS studies, and electrochemical measurements. The likelihood of N participating in the active sites is low because the catalytic performance does not depend on pyridinic and graphitic N.
- This article is part of the themed collection: 2015 Journal of Materials Chemistry A Hot Papers

 Please wait while we load your content...
Please wait while we load your content...