CO2-tolerant oxygen-permeable perovskite-type membranes with high permeability†
Abstract
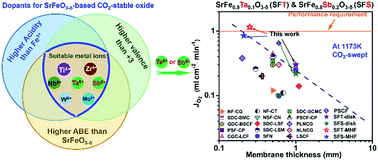
There is a desire for CO2-tolerant oxygen-permeable membranes for CO2 capture based on the oxyfuel process. Here we report a general doping strategy for developing CO2-tolerant SrFeO3−δ-based oxygen-permeable membranes. To combine excellent CO2 tolerance and high permeability, two novel CO2-tolerant oxygen-permeable membranes of SrFe0.9Ta0.1O3−δ (SFT) and SrFe0.8Sb0.2O3−δ (SFS) have been developed based on this doping strategy. Both SFT and SFS oxides possessed high phase stability especially in a pure CO2 atmosphere for 96 h at 1173 K. The high CO2-tolerant properties were mainly associated with high acidity, high valence and appropriate ionic radius of the Ta or Sb cation and high average metal bond energy of SFT or SFS oxide. Both SFT and SFS disk membranes (1 mm-thick) with low oxygen permeation activation energies exhibited high oxygen permeation fluxes of 0.3 and 0.22 ml min−1 cm−2, respectively, which were unchanged during the long-term operation (130 h) under air/CO2 gradient at 1173 K. Furthermore, the highest oxygen permeation flux of 1.15 ml min−1 cm−2 through the SFT multichannel hollow fiber (MHF) membrane at 1173 K under air/CO2 gradient can meet the requirement of commercial application in the oxyfuel process. The present results would give guidance for the design of CO2-tolerant SF-based membranes.

 Please wait while we load your content...
Please wait while we load your content...