Unseeded hydroxide-mediated synthesis and CO2 adsorption properties of an aluminosilicate zeolite with the RTH topology†
Abstract
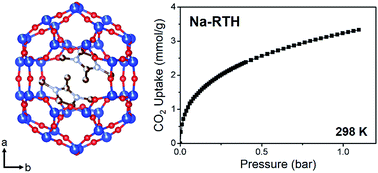
We have synthesized an aluminosilicate RTH-type zeolite with Si/Al = 10 using 1,2,3-trimethylimidazolium (123TMI+) as an organic structure-directing agent (OSDA) together with Na+ or K+ in hydroxide media and without the use of seed crystals. The zeolite obtained is characterized by a cuboid morphology made of very small ill-defined crystallites, largely different from the plank-like morphology typically observed for RTH-type zeolite crystals thus far. More interestingly, we show experimental evidence demonstrating that two 123TMI+ ions are located within each [46586484] cavity of the RTH framework, forming antiparallel dimers, as found by Rietveld refinement. When hydrothermally aged at 1023 K, Cu-RTH is much less active for NO reduction with NH3 than Cu-SSZ-13, the best catalyst known for this reaction to date. However, while the CO2 uptake (3.2 mmol g−1) on Na-RTH at 298 K and 1.0 bar is lower than that (4.5 mmol g−1) on zeolite Na-Rho, a well-studied small-pore zeolite that selectively adsorbs CO2, it exhibits much faster CO2 sorption kinetics. This renders our RTH zeolite potentially useful as a selective CO2 adsorbent.
- This article is part of the themed collection: 2015 Journal of Materials Chemistry A Hot Papers

 Please wait while we load your content...
Please wait while we load your content...