Ultrafast palladium diffusion in germanium†
Abstract
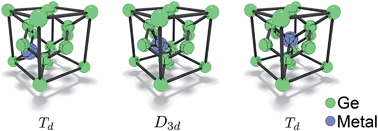
The slow transport of dopants through crystal lattices has hindered the development of novel devices. Typically atoms are contained within deep potential energy wells which necessitates multiple attempts to hop between minimum energy positions. This is because the bonds that constrain atoms are strongest at the minimum positions. As they hop between sites the bonds must be broken, only to re-form as the atoms slide into adjacent minima. Here we demonstrate that the Pd atoms introduced into the Ge lattice behave differently. They retain bonds as the atoms shift across so that at the energy maximum between sites Pd still exhibits strong bonding characteristics. This reduces the energy maximum to almost nothing (a migration energy of only 0.03 eV) and means that the transport of Pd through the Ge lattice is ultrafast. We scrutinize the bonding characteristics at the atomic level using quantum mechanical simulation tools and demonstrate why Pd behaves so differently to other metals we investigated (i.e. Li, Cu, Ag, Pt and Au). Consequently, this fundamental understanding can be extended to systems where extremely rapid diffusion is desired, such as radiation sensors, batteries and solid oxide fuel cells.

 Please wait while we load your content...
Please wait while we load your content...