Phosphorus–carbon nanocomposite anodes for lithium-ion and sodium-ion batteries
Abstract
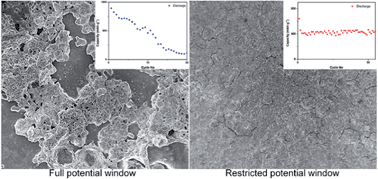
With the expected theoretical capacity of 2596 mA h g−1, phosphorus is considered to be the highest capacity anode material for sodium-ion batteries and one of the most attractive anode materials for lithium-ion systems. This work presents a comprehensive study of phosphorus–carbon nanocomposite anodes for both lithium-ion and sodium-ion batteries. The composite electrodes are able to display high initial capacities of approximately 1700 and 1300 mA h g−1 in lithium and sodium half-cells, respectively, when the cells are tested within a larger potential windows of 2.0–0.01 V vs. Li/Li+ and Na/Na+. The level of demonstrated capacity is underpinned by the storage mechanism, based on the transformation of phosphorus to Li3P phase for lithium cells and an incomplete transformation to Na3P phase for sodium cells. The capacity deteriorates upon cycling, which is shown to originate from disintegration of electrodes and their delamination from current collectors by post-cycling ex situ electron microscopy. Stable cyclic performance at the level of ∼700 and ∼350–400 mA h g−1 can be achieved if the potential windows are restricted to 2.0–0.67 V vs. Li/Li+ for lithium and 2–0.33 vs. Na/Na+ for sodium half-cells. The results are critically discussed in light of existing literature reports.

 Please wait while we load your content...
Please wait while we load your content...