Formation chemistry of perovskites with mixed iodide/chloride content and the implications on charge transport properties†
Abstract
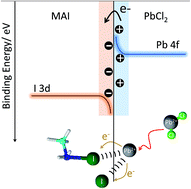
Although Cl-doping is a common technique for achieving high photovoltaic (PV) performance, the Cl content is negligibly small and cannot easily be tuned. Therefore, we herein study the formation chemistry of Cl-doped perovskites by examining the chemical interactions between thermally evaporated MAI and PbCl2 through X-ray photoemission spectroscopy (XPS). We show that PbCl2 is not stable at the MAI/PbCl2 contact surface. The Cl atom readily detaches from the PbCl2, which subsequently initiates electron transition from Pb to MAI. Via thermal-evaporation, a perovskite with a high PbCl2 content can be prepared and examined. We found that the presence of metallic Pb, associated with increased Cl content, can quench the photogenerated exciton in PV devices. By optimizing the ratio of MAI : PbCl2, a perovskite solar cell with ∼6% efficiency was obtained.
- This article is part of the themed collection: Perovskite Solar Cells

 Please wait while we load your content...
Please wait while we load your content...