Synthesis of core–shell Au–Pt nanodendrites with high catalytic performance via overgrowth of platinum on in situ gold nanoparticles†
Abstract
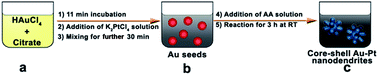
We present a simple and effective strategy for high yield synthesis of well-dispersed, core–shell Au–Pt nanodendrites (CS Au–Pt NDs) via overgrowth of platinum on in situ 5.5 nm gold nanoparticles in water at room temperature. The sizes of the resulting CS Au–Pt NDs are 14 nm, which should be the smallest so far to the best of our knowledge. The average dimensions of the small Pt branches on the Au nanoparticle surfaces are about 2.6 nm × 4.2 nm, which lead to a significantly increased electrochemically active surface area (up to 35.2 m2 g−1). It is found that the morphology of CS Au–Pt NDs is dependent on the reaction conditions such as the incubation time of citrate–HAuCl4 solution, the mixing time of citrate–HAuCl4–K2PtCl4 solution before AA addition, and Pt-to-Au and AA-to-Pt molar ratios. In comparison with commercial Pt black (0.12 A mgPd−1), the resulting Au–Pt5 NDs show a superior catalytic activity towards methanol oxidation (0.45 A mgPd−1) due to the electronic interaction between the Au cores and Pt branches in bimetallic Au–Pt NDs and the high fraction of atomic steps, kinks, and corner atoms on the surfaces of the Pt branches.

 Please wait while we load your content...
Please wait while we load your content...