Polyaniline nanofiber/electrochemically reduced graphene oxide layer-by-layer electrodes for electrochemical energy storage†
Abstract
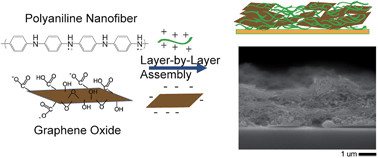
Graphene-containing layer-by-layer (LbL) electrodes are promising for thin film electrochemical energy storage. However, common practice centers on assembly with chemically reduced graphene oxide sheets, which have a tendency to severely aggregate during processing. More direct and facile is the LbL assembly of graphene oxide (GO) sheets and their subsequent electrochemical reduction. Here, we demonstrate porous (void fraction = 0.625) LbL electrodes comprised of electrochemically reduced GO (ERGO) sheets and polyaniline nanofibers (PANI NFs) for use in non-aqueous energy storage systems. Our approach is also promising for deposition onto complex surfaces, as demonstrated here by the successful assembly onto cotton fabric. Both PANI NFs and ERGO sheets store charge, bear conductivity, and provide a highly porous architecture, which facilitates the mass transport of ions. The nature of PANI NF/GO LbL assembly and growth is first presented, which we find to be affected by assembly pH. The confirmation of the electrochemical reduction step is then discussed, followed by the electrochemical performance of the resulting electrodes in a non-aqueous lithium metal battery. Capacity varies from 85 to 184 mA h cm−3 (188 to 461 mA h g−1) at 0.1 A g−1 (electrode mass basis), depending on the electrode thickness. The highest specific energy measured was 1395 mW h g−1 at a specific power of 1590 mW g−1, and the highest specific power was 60 252 mW g−1 at a specific energy of 927 mW h g−1. These results demonstrate that electroactive polyaniline nanofiber/graphene coatings from aqueous layer-by-layer assembly are attainable for energy storage.

 Please wait while we load your content...
Please wait while we load your content...