Natural products triptolide, celastrol, and withaferin A inhibit the chaperone activity of peroxiredoxin I†
Abstract
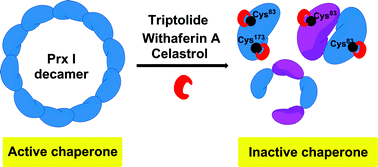
Peroxiredoxin I (Prx I) plays an important role in cancer development and inflammation. It is a dual-functional protein which acts as both an antioxidant enzyme and a molecular chaperone. While there have been intensive studies on its peroxidase activity, Prx I's chaperone activity remains elusive, likely due to the lack of chaperone inhibitors. Here we report that natural product triptolide selectively inhibits the chaperone activity of Prx I, but not its peroxidase activity. Through direct interaction with corresponding cysteines, triptolide triggers dissociation of high-molecular-weight oligomers of Prx I, and thereby inhibits its chaperone activity in a dose-dependent manner. We have also identified celastrol and withaferin A as novel Prx I chaperone inhibitors that are even more potent than triptolide in the chaperone activity assay. By revealing the exact molecular mechanisms of interaction and inhibition, the current study provides the first Prx I chaperone inhibitors as promising pharmacological tools for modulating and dissecting the chaperone function of Prx I.

 Please wait while we load your content...
Please wait while we load your content...