IspH–RPS1 and IspH–UbiA: “Rosetta stone” proteins†
Abstract
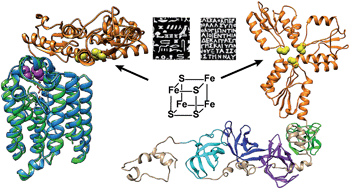
The protein IspH, (E)-1-hydroxy-2-methyl-but-2-enyl 4-diphosphate (HMPPP) reductase, is an essential 4Fe–4S cluster-containing protein in the methylerythritol phosphate pathway for isoprenoid biosynthesis. Using a sequence similarity network we found that there are >400 IspH proteins that are about twice as large as most of the IspHs studied to date since their IspH domains are fused to either the ribosomal protein S1 (RPS1), or to a UbiA (4-hydroxybenzoate octaprenyltransferase)-like protein. Many of the IspH–RPS1 proteins are present in anaerobes found in the human gut and some, such as Clostridium botulinum, C. tetani and Fusobacterium nucleatum, are pathogens. The IspH–UbiAs are all found in sulfate-reducing anaerobes. The IspH domains in IspH–RPS1 are fused to 4 and in a few cases 6 tandem repeats in RPS1 that, in most organisms, bind to mRNA or form part of the bacterial ribosome. Mutants in which the four RPS1 domains were sequentially eliminated had similar IspH activity as wild-type protein, indicating they are not essential for IspH catalysis. Overall, the results are of interest since they represent the first isolation of a catalytically active IspH–RPS1, as well as the identification of IspH–UbiA hybrids, two “Rosetta stone” proteins that are likely to be functionally related—IspH producing the isoprenoids required for a UbiA-like prenyltransferase; the IspH–RPS1 hybrids, perhaps, being involved in the stringent response or as Fe/O2 sensors.


 Please wait while we load your content...
Please wait while we load your content...