Selective acceleration of disfavored enolate addition reactions by anion–π interactions†
Abstract
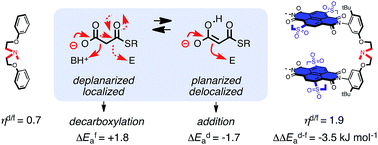
In chemistry and biology, cation–π interactions contribute significantly to many important transformations. In sharp contrast, reactions accomplished with support from the complementary anion–π interactions are essentially unknown. In this report, we show that anion–π interactions can determine the selectivity of the enolate chemistry of malonate half thioesters. Their addition to enolate acceptors is central in natural product biosynthesis but fails without enzymes because non-productive decarboxylation dominates. The newly designed and synthesized anion–π tweezers invert this selectivity by accelerating the disfavored and decelerating the favored process. The discrimination of anionic tautomers of different planarization and charge delocalization on π-acidic surfaces is expected to account for this intriguing “tortoise-and-hare catalysis.” Almost exponentially increasing selectivity with increasing π acidity of the catalyst supports that contributions from anion–π interactions are decisive.

 Please wait while we load your content...
Please wait while we load your content...