A soluble molecular variant of the semiconducting silicondiselenide†
Abstract
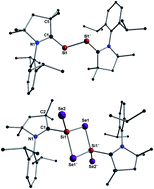
Silicondiselenide is a semiconductor and exists as an insoluble polymer (SiSe2)n which is prepared by reacting elemental silicon with selenium powder in the temperature range of 400–850 °C. Herein, we report on the synthesis, isolation, and characterization of carbene stabilized molecular silicondiselenide in the form of (cAAC)2Si2Se4 (3) [cAAC = cyclic alkyl(amino)carbene]. 3 is synthesized via reaction of diatomic silicon(0) compound (cAAC)2Si2 (2) with black selenium powder at −78 °C to room temperature. The intensely orange colored compound 3 is soluble in polar organic solvents and stable at room temperature for a month under an inert atmosphere. 3 decomposes above 245 °C. The molecular structure of 3 has been confirmed by X-ray single crystal diffraction. It is also characterized by UV-vis, IR, Raman spectroscopy and mass spectrometry. The stability, bonding, and electron density distributions of 3 have been studied by theoretical calculations.

 Please wait while we load your content...
Please wait while we load your content...