Enhancing H2 evolution performance of an immobilised cobalt catalyst by rational ligand design†
Abstract
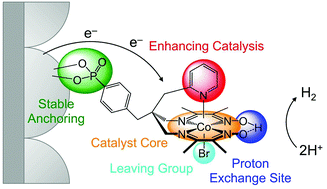
The catalyst [CoIIIBr((DO)(DOH)(4-BnPO3H2)(2-CH2py)pn)]Br, CoP3, has been synthesised to improve the stability and activity of cobalt catalysts immobilised on metal oxide surfaces. The CoP3 catalyst contains an equatorial diimine–dioxime ligand, (DOH)2pn = N2,N2′-propanediyl-bis(2,3-butanedione-2-imine-3-oxime), with a benzylphosphonic acid (4-BnPO3H2) group and a methylpyridine (2-CH2py) ligand covalently linked to the bridgehead of the pseudo-macrocyclic diimine–dioxime ligand. The phosphonic acid functionality provides a robust anchoring group for immobilisation on metal oxides, whereas the pyridine is coordinated to the Co ion to enhance the catalytic activity of the catalyst. Electrochemical investigations in solution confirm that CoP3 shows electrocatalytic activity for the reduction of aqueous protons between pH 3 and 7. The metal oxide anchor provides the catalyst with a high affinity for mesostructured Sn-doped In2O3 electrodes (mesoITO; loading of approximately 22 nmol cm−2) and the electrostability of the attached CoP3 was confirmed by cyclic voltammetry. Finally, immobilisation of the catalyst on ruthenium-dye sensitised TiO2 nanoparticles in aqueous solutions in the presence of a hole scavenger establishes the activity of the catalyst in this photocatalytic scheme. The advantages of the elaborate catalyst design in CoP3 in terms of stability and catalytic activity are shown by direct comparison with previously reported phosphonated Co catalysts. We therefore demonstrate that rational ligand design is a viable route for improving the performance of immobilised molecular catalysts.
- This article is part of the themed collection: Global Energy Challenges: Hydrogen Energy

 Please wait while we load your content...
Please wait while we load your content...