Effects of conformational symmetry in conjugated side chains on intermolecular packing of conjugated polymers and photovoltaic properties†
Abstract
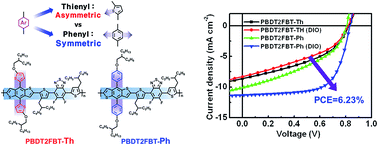
Introducing conjugated side chains onto the backbone of two-dimensionally (2D) conjugated polymers has been utilized for tuning the optoelectronic characteristics of the polymer and the morphological properties of organic photovoltaics. To investigate the effects of conformational symmetry of conjugated side chains, two benzo[1,2-b:3,4-b′]dithiophene (BDT)-based derivatives, one with the asymmetric alkoxythienyl (Th) side chain and the other with the symmetric alkoxyphenyl (Ph) side chain, were synthesized as donor units and copolymerized with fluorinated benzothiadiazole (2FBT). These two side chains were selected for the distinct differences between their structures, and were found to affect the intrinsic characteristics of these BDT polymers. The introduction of the symmetric conjugated side chain to the conjugated backbone of the polymer was observed to improve both light harvesting and the charge carrier mobility, apparently by increasing the extent of packing between the polymer chains. Power conversion efficiency (PCE) values of photovoltaic devices fabricated using these conjugated polymers were strongly related to the light absorbance and crystallinity in a film of the blend of polymer and [6,6]-phenyl C71-butyric acid methyl ester (PC71BM). PBDT2FBT-Ph showed effective light absorption, an optimum morphology that we argue is due to the symmetry of the conjugated Ph side chain, and a maximum PCE of 6.23%, with an open-circuit voltage of 0.83 V, a short-circuit current density of 11.33 mA cm−2, and a fill factor of 66.3%. These results demonstrate that symmetric conjugated side chains are promising groups to produce 2D-conjugated polymers for high-performance photovoltaics. This systematic study of side chain engineering provides a valuable strategy to synthesize 2D conjugated polymers and to achieve high PCE values in organic photovoltaics.

 Please wait while we load your content...
Please wait while we load your content...