Involvement of covalent interactions in the mode of action of PPARβ/δ antagonists†
Abstract
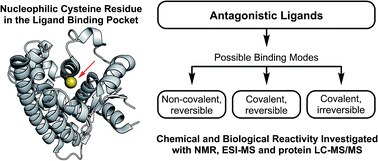
A broad range of chemical structures modulate the inductive and repressive transcriptional regulation of the peroxisome proliferator-activated receptor β/δ (PPARβ/δ). In order to shed light on mechanistic differences in the modes of action of three classes of the reported PPARβ/δ antagonists, an investigation into their in vitro biological and chemical reactivities, with particular focus on covalent reactivity, was undertaken. The results reported here, substantiate the covalent modification of Cys249 as a part of the mode of action of the 5-trifluoromethyl-2-sulfonylpyridine class of antagonists. In contrast, GSK0660 does not appear to be a covalently binding antagonistic ligand. Additionally, we demonstrate the electrophilic nature of the recently published antagonist DG172 towards thiolates, although a covalent adduct with PPARβ/δ is not detected in our experiments.

 Please wait while we load your content...
Please wait while we load your content...