Dye-sensitization of boron-doped diamond foam: champion photoelectrochemical performance of diamond electrodes under solar light illumination†
Abstract
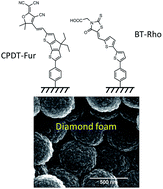
Diamond foams composed of hollow spheres of polycrystalline boron-doped diamond are chemically modified with two donor–acceptor type molecular dyes, BT-Rho and CPDT-Fur, and tested as electrode materials for p-type dye-sensitized solar cells with an aqueous electrolyte solution containing methyl viologen as a redox mediator. Reference experiments with flat polycrystalline diamond electrodes evidence full blocking of the methyl viologen redox reaction by these dyes, whereas only partial blocking is observed for the diamond foams. This is ascribed to sp2-carbon impurities in the foam, viz. trans-polyacetylene and graphite-like carbon. Cathodic photocurrents under solar light illumination are about 3 times larger on foam electrodes compared to flat diamond. Long-term (1–2 days) illumination of the sensitized foam electrodes with chopped light at 1 sun intensity causes an increase of the cathodic photocurrent density to ca. 15–22 μA cm−2. These photocurrent densities represent the largest values reported so far for dye-sensitized diamond electrodes. The photoelectrochemical activation of the sensitized diamond electrodes is accompanied with characteristic changes of the dark voltammogram of the MV2+/MV+ redox couple and with gradual changes of the IPCE spectra.

 Please wait while we load your content...
Please wait while we load your content...