Synthesis and characterization of Ni doped SnO2 microspheres with enhanced visible-light photocatalytic activity†
Abstract
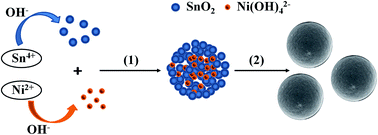
Nickel-doped tin dioxide (NDT) microspheres with various doping levels were prepared by hydrothermal synthesis and characterized by using X-ray powder diffraction, scanning electron microscopy, transmission electron microscopy, high-resolution transmission electron microscopy, energy-dispersive spectroscopy analysis, Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and UV-vis absorption spectroscopy. The formation mechanism of NDT microspheres was discussed. All samples prepared with different Sn/Ni ratios shows higher photocatalytic activity than that of pure SnO2 and pure NiO under visible light irradiation (λ > 420 nm). The photocatalytic activity is closely related to the composition of as-prepared photocatalysts. It is noted that the NDT microspheres with the doping content of 33.3 mol% exhibit the highest photoactivity. Moreover, this catalyst showed improved stability, and the activity did not weaken significantly after four recycles. The higher activity of NDT microspheres may be attributed to the combined effect of its demonstrated doping levels and appropriate band gap. And the doping of Ni2+ may facilitate the generation of electron and hole pairs and inhibit their recombination rate by acting as temporary trapping sites of photoinduced electrons.

 Please wait while we load your content...
Please wait while we load your content...