Structural elucidation of thermolysis products of methyl N-methyl-N-nitrosoanthranilate
Abstract
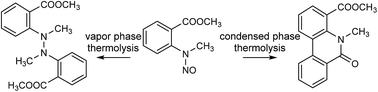
Although it is common knowledge that N-nitroso compounds are thermally (and otherwise chemically) labile, little or nothing is known about the specific reactions that occur during thermal treatment of a compound possessing this functionality. Methyl N-methyl-N-nitrosoanthranilate was found to undergo complete thermal degradation under gas chromatographic (GC–MS) conditions yielding a major unidentified coupling product and methyl N-methylanthranilate. In an attempt to elucidate the structure of the formed degradation product, a preparative scale thermolysis in an evacuated vessel at 220 °C of the nitroso compound was carried out. A chromatographic separation of the thermolysate, followed by GC–MS and NMR (and other spectral techniques) analyses enabled the identification of in total 46 different products. Among them a novel coupling product, methyl 5-methyl-6-oxo-(5H)-phenanthridine-4-carboxylate, was identified and fully spectrally characterized. Interestingly, the initially detected coupling product that formed under GC conditions, tentatively identified as dimethyl 2,2′-(1,2-dimethylhydrazine-1,2-diyl)dibenzoate, was not detected in the thermolysate. A careful consideration of the structures of the identified thermolysate constituents led us to propose major thermolysis pathways of methyl N-methyl-N-nitrosoanthranilate both in condensed and vapor phases. Generally, the identified products could be classified as those arising from fission of the N–NO bond or rather unexpectedly, the Ar–NNO bond, i.e. products related to anthranilic and benzoic acids, respectively. The latter represents a novel chemical transformation up to now unreported and unutilized. The structural diversity of the identified products and the noted marked differences between vapor and condensed phases point to the possible synthetic utility of thermolysis of carefully designed N-nitroso compounds.

 Please wait while we load your content...
Please wait while we load your content...