Solvent determines the formation and properties of metal–organic frameworks†
Abstract
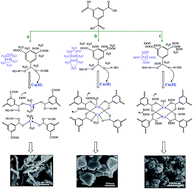
The formation of a water-sensitive metal–organic framework (MOF), Cu3(BTC)2 (BTC = 1,3,5-benzenetricarboxylate), in a water/ethanol solvent system was studied systematically. The X-ray diffraction results prove that the MOF cannot form in pure water or in a water/ethanol mixture with a small amount of ethanol. As the ethanol content exceeds 30 vol%, a crystalline MOF can be obtained. The scanning electron microscope images of the as-synthesized MOFs show the formation of MOF nanoparticles with an average size of 20–300 nm. The MOF particle size decreased with increasing ethanol content in the mixed solvent. The FT-IR spectra further support that the MOF formation occurs in water/ethanol mixtures with ethanol volume ratios higher than 30 vol%. Thermogravimetric analysis showed that the MOF is stable up to 300 °C. Moreover, the FT-IR spectra and thermogravimetric analysis gave consistent information on the solvent amount entrapped in the MOF pores. The porosity of the MOFs was determined using a N2 adsorption–desorption method. When the ethanol volume ratio reached 75%, the largest SBET value of 1067 m2 g−1 and Vt value of 0.52 cm3 g−1 were obtained. The possible mechanism for MOF formation in water/ethanol solvent systems and the dependence of the MOF size on the solvent composition was discussed from the view of hydrogen bond strength between solvent molecules and the ligands in different water/ethanol solvent systems.

 Please wait while we load your content...
Please wait while we load your content...