Structure, absolute configuration, and conformational study of resorcylic acid derivatives and related congeners from the fungus Penicillium brocae†
Abstract
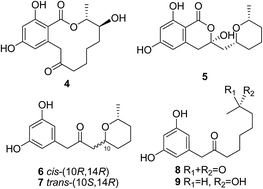
In addition to three known resorcylic acid derivatives of the (14R) series (1–3), six new compounds including one new resorcylic acid derivative (13-hydroxydihydroresorcylide, 4) and five new loop-opened resorcylic acid-related congeners including brocapyrone A (5) and brocaketones A–D (6–9), were isolated and identified from the culture extract of a marine mangrove-derived endophytic fungus Penicillium brocae MA-192. Conformational analysis and TDDFT-ECD calculations of 1, 3, and 4 were carried out to confirm the (14R) absolute configuration and identify the solution conformers and factors that govern the correlation between stereochemistry and signs of the characteristic ECD bands. The single crystal X-ray of 3 and 5 allowed applying the solid-state TDDFT-ECD approach for 3 to compare the structures of solution and solid-state conformers and correlate the n–π* ECD transition of 5 with the helicity and absolute configuration of the dihydroisocoumarin moiety. All compounds were evaluated for antioxidant activity against DPPH and ABTS radicals.

 Please wait while we load your content...
Please wait while we load your content...