Visible-light-driven photocatalytic H2 evolution from aqueous suspensions of perylene diimide dye-sensitized Pt/TiO2 catalysts
Abstract
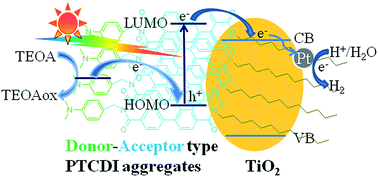
TiO2 is among the most important semiconductor photocatalysts for solar light-driven H2 production from photocatalytic water reduction. Commercialized mixed-phase Degussa P25 nanoparticles have been demonstrated as the most popular TiO2. However, its superiority cannot be exerted in the visible-light region. In this work, molecular aggregates of perylene tetracarboxylic diimide (PTCDI, a kind of air-stable n-type organic semiconductor bearing an electron-rich phenylamino moiety and a dodecyl side-chain) were employed as a sensitizer to combine with Pt/TiO2 nanoparticles via solution processing. The PTCDI/Pt/TiO2 composites thus fabricated exhibit a broad visible-light response and excellent photochemical stability. The initial intramolecular charge transfer of PTCDI and its energy level being well matched to TiO2, ensure effective charge transfer among the PTCDI/Pt/TiO2 composite. When 25 mg of the composite powders are suspended in aqueous solution (100 mL) containing 10 mL of triethanolamine as a sacrificial electron donor, stable H2 evolution with an activity of ∼0.075 μmol h−1 is achieved under visible-light (λ ≥ 420 nm) irradiation. Both intramolecular electron transfer from the electron rich 4-dimethylaminobenzyl group to the perylene core of PTCDI and the intermolecular charge transfer from PTCDI to TiO2 are key to the effective visible-light photoactivity.

 Please wait while we load your content...
Please wait while we load your content...