Synthesis and properties of polyesters derived from renewable eugenol and α,ω-diols via a continuous overheating method†
Abstract
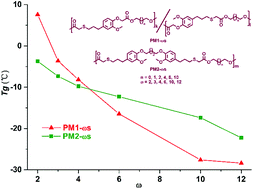
Two eugenol-based aromatic dimethyl terephthalate (DMT)-like monomers were prepared via a thiol–ene click reaction and a subsequent nucleophilic substitution reaction with methyl chloroacetate or 1,4-dibromobutane. Two series of thermoplastic polyesters derived from renewable eugenol and linear aliphatic α,ω-diols HO–(CH2)n–OH (n = 2, 3, 4, 6, 10, 12) were successfully synthesized. These prepared polyesters have weight-average molecular weights in the range of 18 500–90 500 g mol−1, and polydispersity indexes (PDIs) between 1.8 and 2.2. Their chemical structures were all accurately characterized by 1H NMR spectroscopy, 13C NMR spectroscopy and FTIR spectroscopy. The random microstructures of the synthetic polyesters were also explored by 13C NMR spectroscopy. The obtained polyesters all exhibit a thermal stability above 330 °C. More importantly, the thermal stability, the maximum degradation rate and the residue weight are intimately associated with the length of the linear aliphatic α,ω-diol. Their thermo-mechanical properties were studied by differential scanning calorimetry (DSC) and dynamic mechanical analysis (DMA). The length of the linear aliphatic α,ω-diol crucially influences the glass transition temperature (Tg). With the gradual increase in the α,ω-diol length, the Tg of the synthesized polyester exhibits a tendency to decrease. The polyesters are all amorphous materials at room temperature, with Tg values ranging from −28.4 to 7.6 °C. The incorporation of aromatic eugenol into the polyester chains reduces the crystallinity significantly. The Young's modulus and ultimate strength are in the range of 1.2–6.9 MPa and 0.96–3.37 MPa, respectively. On the contrary, the elongation at break reaches up to 840–1000%, indicating the excellent viscosity properties for such unmanageable viscous materials.

 Please wait while we load your content...
Please wait while we load your content...