Controlled micellar disassembly of photo- and pH-cleavable linear-dendritic block copolymers†
Abstract
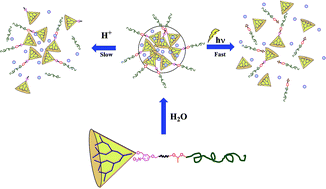
A linear-dendritic block copolymer with both photo- and pH-cleavable linkages at the junction between the hydrophobic dendron and hydrophilic linear polymer was synthesised. The photocleavable o-nitrobenzyl group attached to alkyne was introduced at the focal point of polyester dendron and acid-degradable acetal linkage with terminal azide was introduced at one end of linear polyethylene glycol. The two segments were connected using click chemistry. The copolymer was found to self-assemble into micelle-like aggregates in dilute aqueous solution. Stimuli-responsive disassembly of the aggregates was studied by monitoring fluorescence emission of the encapsulated hydrophobic dye and dynamic light scattering (DLS). Products of acetal cleavage at acidic pH were analyzed by gel permeation chromatography and NMR spectroscopy. The photo and pH stimuli were applied separately as well as simultaneously to study the rate of dye release. The synergistic effect of the two stimuli on dye release was demonstrated and the non-cytotoxic nature of the polymer was shown using the MTT assay. Cell uptake of DOX-loaded micelles and photo-induced release of the drug resulting in significantly higher cytotoxicity than by free DOX was demonstrated using MDA-MB-231 cells.

 Please wait while we load your content...
Please wait while we load your content...