Characterization of photo-intermediates in the photo-reaction pathways of a bacteriorhodopsin Y185F mutant using in situ photo-irradiation solid-state NMR spectroscopy†
Abstract
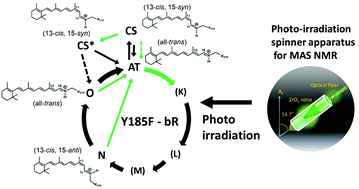
Photo-reaction pathways of a bacteriorhodopsin Y185F mutant were examined using in situ photo-irradiation solid-state NMR spectroscopy. 13C CP MAS NMR spectra were recorded at −40 °C in the dark (D1), under irradiation with 520 nm light (L1), subsequently in the dark (D2), and again under irradiation with 520 nm light (L2). In the process from D1 to L1, the 13-cis, 15-syn (CS; bR548) state changed to a CS*- (13-cis, 15-syn) intermediate, which was highly stable at −40 °C, and the all-trans (AT; bR568) state transformed to an N-intermediate. Under the D2 conditions, the N-intermediate transformed to an O-intermediate, which was highly stable at −40 °C in the dark. During subsequent irradiation with 520 nm light (L2), the O-intermediate transformed to the N-intermediate through the AT state, whereas the CS*-intermediate did not change. The CS*-intermediate was converted to the AT state (or O-intermediate) after the temperature was increased to −20 °C. Upon subsequent increase of the temperature to 20 °C, the AT state (or O-intermediate) was converted to the CS state until reaching equilibrium. In this experiment, the chemical shift values of [20-13C, 14-13C]retinal provided the 13C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and 15C
C and 15C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N configurations, respectively. From these data, the configurations of the AT and CS states and the CS*-, N-, and O-intermediates were determined to be (13-trans, 15-anti), (13-cis, 15-syn), (13-cis, 15-syn), (13-cis, 15-anti), and (13-trans, 15-anti), respectively. 13C NMR signals of the CS*- and O-intermediates were observed for the first time for the Y185F bR mutant by in situ photo-irradiation solid-state NMR spectroscopy and the configuration of the CS*-intermediate was revealed to be significantly twisted from that of the CS state although both were assigned as (13-cis, 15-syn) configurations.
N configurations, respectively. From these data, the configurations of the AT and CS states and the CS*-, N-, and O-intermediates were determined to be (13-trans, 15-anti), (13-cis, 15-syn), (13-cis, 15-syn), (13-cis, 15-anti), and (13-trans, 15-anti), respectively. 13C NMR signals of the CS*- and O-intermediates were observed for the first time for the Y185F bR mutant by in situ photo-irradiation solid-state NMR spectroscopy and the configuration of the CS*-intermediate was revealed to be significantly twisted from that of the CS state although both were assigned as (13-cis, 15-syn) configurations.
- This article is part of the themed collection: 16th International Conference on Retinal Proteins

 Please wait while we load your content...
Please wait while we load your content...