Metallated porphyrin based porous organic polymers as efficient electrocatalysts†
Abstract
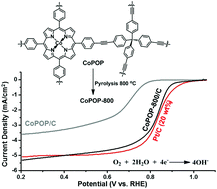
Developing efficient, stable and low-cost catalysts for Oxygen Reduction Reaction (ORR) is of great significance to many emerging technologies including fuel cells and metal–air batteries. Herein, we report the development of a cobalt(II) porphyrin based porous organic polymer (CoPOP) and its pyrolyzed derivatives as highly active ORR catalysts. The as-synthesized CoPOP exhibits high porosity and excellent catalytic performance stability, retaining ∼100% constant ORR current over 50 000 s in both alkaline and acidic media. Pyrolysis of CoPOP at various temperatures (600 °C, 800 °C, and 1000 °C) yields the materials consisting of graphitic carbon layers and cobalt nanoparticles, which show greatly enhanced catalytic activity compared to the as-synthesized CoPOP. Among them, CoPOP-800/C pyrolyzed at 800 °C shows the highest specific surface area and ORR activity, displaying the most positive half-wave potential (0.825 V vs. RHE) and the largest limited diffusion current density (5.35 mA cm−2) in an alkaline medium, which are comparable to those of commercial Pt/C (20 wt%) (half-wave potential 0.829 V vs. RHE, limited diffusion current density 5.10 mA cm−2). RDE and RRDE experiments indicate that CoPOP-800/C directly reduces molecular oxygen to water through a 4-e− pathway in both alkaline and acidic media. More importantly, CoPOP-800/C exhibits excellent durability and methanol-tolerance under acidic and alkaline conditions, which surpass the Pt/C (20 wt%) system.

 Please wait while we load your content...
Please wait while we load your content...