Ionic liquid self-combustion synthesis of BiOBr/Bi24O31Br10 heterojunctions with exceptional visible-light photocatalytic performances†
Abstract
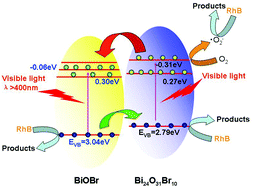
Heterostructured BiOBr/Bi24O31Br10 nanocomposites with surface oxygen vacancies are constructed by a facile in situ route of one-step self-combustion of ionic liquids. The compositions can be easily controlled by simply adjusting the fuel ratio of urea and 2-bromoethylamine hydrobromide (BTH). BTH serves not only as a fuel, but also as a complexing agent for ionic liquids and a reactant to supply the Br element. The heterojunctions show remarkable adsorptive ability for both the cationic dye of rhodamine B (RhB) and the anionic dye of methylene orange (MO) at high concentrations, which is attributed to the abundant surface oxygen vacancies. The sample containing 75.2% BiOBr and 24.8% Bi24O31Br10 exhibits the highest photocatalytic activity. Its reaction rate constant is 4.0 and 9.0 times that of pure BiOBr in degrading 50 mg L−1 of RhB and 30 mg L−1 of MO under visible-light (λ > 400 nm) irradiation, respectively, which is attributed to the narrow band gap and highly efficient transfer efficiency of charge carriers. Different photocatalytic reaction processes and mechanisms over pure BiOBr and heterojunctions are proposed.
- This article is part of the themed collection: Editor’s Choice: Nanomaterials for catalysis

 Please wait while we load your content...
Please wait while we load your content...