Kinetically-controlled growth of cubic and octahedral Rh–Pd alloy oxygen reduction electrocatalysts with high activity and durability†
Abstract
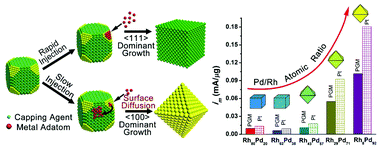
Rh is a promising candidate as an indispensible component in bimetallic catalysts due to its unique capability to resist against the aggressive corrosion from the reaction medium. However, Rh has a very strong oxygen binding ability and is generally not suitable for the oxygen reduction reaction (ORR). Here, we have demonstrated shape-controlled synthesis of Rh–Pd alloy nanocrystals with high activity and durability for ORR by retarding the reaction kinetics at an ultra-slow injection rate of metal salts using a syringe pump. Under precise control of sluggish reaction kinetics, Pd followed a preferential overgrowth along the <100> direction, whereas the growth behavior of Rh was dominant along the <111> direction. These different kinetically-controlled growth behaviors associated with Rh and Pd were essential for achieving the shape transition between the cube and the octahedron of their alloys. The Rh8Pd92 alloy octahedra exhibited the highest mass activity with a value of 0.18 mA μg−1 in terms of the equivalent Pt cost, and were two-fold higher than that of commercial Pt/C. Significantly, all Rh–Pd alloy nanocrystals were highly stable with only less than 25% loss in mass activity after 30 000 CV cycles in O2 saturated acid solution compared to ∼56% loss of the commercial Pt/C (E-TEK). Indeed, the mass activity of Rh8Pd92 was 3.3 times higher than that of commercial Pt/C after the accelerated stability test (ADT). This improvement in activity and durability may arise possibly from synergistic effects between the facet and the surface composition.

 Please wait while we load your content...
Please wait while we load your content...