Electrochemical characterization of hydroquinone derivatives with different substituents in acetonitrile
Abstract
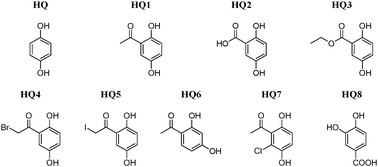
The effect of carbonyl groups in the ortho position with respect to a hydroxyl group on the electrochemical oxidation of hydroquinones in acetonitrile is studied. The electrochemical response of hydroquinone on a glassy carbon electrode in 0.1 M tetrabutylammonium perchlorate was investigated in detail by voltammetry and coulometry. From these experiments, the oxidation potential was shifted to more positive values with respect to hydroquinone due to the presence of electron withdrawing groups bonded to the aromatic ring. For all compounds a diffusional behavior was observed, and the diffusion coefficient (D) of substituted hydroquinones was calculated showing higher values than found for unsubstituted hydroquinone. Theoretical calculations were carried out to gain insights into the intramolecular hydrogen bond present in these molecules affecting their electrochemical behavior. Relevant theoretical data are optimized geometrical parameters, HOMO energy, condensed radical Fukui functions (f°), natural charges, Wiberg bond orders (WBO), stabilization energies caused by electron transfer, and hyperconjugation stabilization energies from the NBO analysis. In most cases, the calculations show good agreement with experimental 1H-NMR data and support the electrochemical results.

 Please wait while we load your content...
Please wait while we load your content...