Speciation of metal-based nanomaterials in human serum characterized by capillary electrophoresis coupled to ICP-MS: a case study of gold nanoparticles†
Abstract
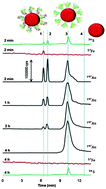
The development and optimization of a versatile analytical system for the speciation analysis of metal-containing nanoscale materials in blood serum is reported herein. Based on capillary electrophoresis (CE) interfaced with inductively coupled plasma mass spectrometry (ICP-MS), the method was shown to be feasible to investigate the interactions between serum proteins and gold nanoparticles of potential medicinal use, which are their first and foremost occurrence upon their entry into the circulatory system. To improve the separation resolution between the intact nanoparticles and different protein conjugates, the CE system was optimized with an emphasis on compatibility with physiological conditions, avoiding aggregation effects, and analyte recovery. Optimization allowed also for acquiring the acceptable figures of merit such as migration time and peak area precision of 1.0–6.4% and 2.4–6.9%, respectively, detection limits in the range of 0.8–1.0 μg L−1 Au, and capillary recoveries on the order of 86–97%, depending on the nanoparticle size and conjugate type. We sytematically investigated the role of size in mediating protein adsorption to gold nanoparticles in a real-serum environment. At the initial stage of surface coating, the speciation of smaller particles (5 and 10 nm) was found to be dominated by albumin, transferrin (both in apo- and holo-form) playing the secondary role in developing the protein corona. For 20 and 50 nm nanoparticles, the contribution of transferrin is initially comparable; however, with time it becomes replaced by albumin. The time of attaining equilibrium adsorption is also a function of particle size but for the whole size range investigated, albumin is the only equilibrium binding partner. These principal findings prove that for metal-based nanomaterials in general, serum protein conjugates could be variable in composition depending on the protein abundance and binding affinity, as well as the residence time in the bloodstream.

 Please wait while we load your content...
Please wait while we load your content...