An infrared spectroscopic study of the nature of zinc carboxylates in oil paintings†
Abstract
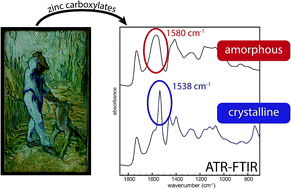
The formation of metal soaps is a major problem for oil paintings conservators. The complexes of either lead or zinc and fatty acids are the product of reactions between common pigments and the oil binder, and they are associated with many types of degradation that affect the appearance and stability of oil paint layers. Fourier transform infrared spectroscopy (FTIR) reveals that a paint sample from The Woodcutter (after Millet) by Vincent van Gogh contains two distinct zinc carboxylate species, one similar to crystalline zinc palmitate and one that is characterized by a broadened asymmetric stretch COO− band shifted to 1570–1590 cm−1. This observation has been made in many paintings. Although several hypotheses exist to explain the shifted broad carboxylate band, these were not supported by experimental evidence. In this paper, experiments were carried out to characterize the second zinc carboxylate type. It is shown that neither variations in the composition of zinc soaps (i.e. zinc soaps containing mixtures of fatty acids or metals) nor fatty acids adsorbed on pigment surfaces are responsible for the second zinc carboxylate species. X-Ray diffraction (XRD) and FTIR analysis indicate that the broad COO− band represents amorphous zinc carboxylates. These species can be interpreted as either non-crystalline zinc soaps or zinc ions bound to carboxylate moieties on the polymerized oil network, a system similar to ionomers. These findings uncover an intermediate stage of metal soap-related degradation of oil paintings, and lead the way to improved methods for the prevention and treatment of oil paint degradation.

 Please wait while we load your content...
Please wait while we load your content...