Gold(i)-catalysed dehydrative formation of ethers from benzylic alcohols and phenols†
Abstract
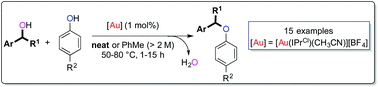
We report the cross-dehydrative reaction of two alcohols to form unsymmetrical ethers using NHC–gold(I) complexes (NHC = N-heterocyclic carbene). Our progress in developing this reaction into a straightforward procedure is discussed in detail. The optimised methodology proceeds under mild reaction conditions and produces water as the sole by-product. The synthetic utility of this environmentally benign methodology is exemplified by the formation of a range of new ethers from readily available phenols bearing electron withdrawing substituents and secondary benzylic alcohols with various substituents. Finally, we present experimental results to account for the chemoselectivity obtained in these reactions.

 Please wait while we load your content...
Please wait while we load your content...