Conversion of cellulose to lactic acid catalyzed by erbium-exchanged montmorillonite K10†
Abstract
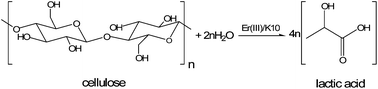
Various erbium ion-exchanged montmorillonite K10 materials were prepared by an ion exchange method and were found to act as efficient solid acid catalysts. The catalytic materials synthesized in this work were characterized using a combination of X-ray fluorescence spectroscopy, N2 adsorption, powder X-ray diffraction, Fourier transform infrared spectroscopy (FT-IR), inductively coupled plasma optical emission spectroscopy, X-ray photoelectron spectroscopy and NH3 temperature-programmed desorption, as well as by FT-IR spectra analysis following pyridine adsorption. These catalysts were also evaluated with regard to the hydrothermal conversion of cellulose to lactic acid. Lactic acid yields as high as 67.6% were obtained when reacting 0.3 g cellulose, 0.1 g catalyst and 30 mL water at 240 °C under 2 MPa N2 for 30 min. Upon recycling of the catalyst, the lactic acid yields decreased from 67.6 to 58.7 to 55.9% during the first, second and third trials. Beginning with the second trial the catalyst behaved as a true heterogeneous catalyst for the conversion of cellulose to lactic acid. The observed decreases in catalytic activity during recycling could be due to a combination of erbium ion leaching, deposition of carbon species in pores and partial structural changes in the catalyst.

 Please wait while we load your content...
Please wait while we load your content...