Abstract
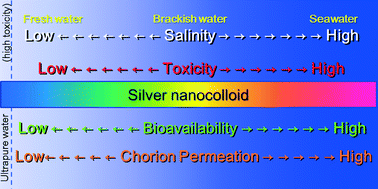
To investigate the effects of salinity on the toxicity of silver nanocolloids (SNCs, 28.4 nm in diameter) in aquatic environments (freshwater, brackish water, and seawater), we exposed 15 medaka eggs in triplicate to SNCs at 10 mg L−1 in different salinities of embryo-rearing medium (ERM) (1×, 5×, 10×, 15×, 20×, and 30×) until hatching (1× ERM and 30× ERM have osmotic pressures equivalent to freshwater and seawater, respectively). With increasing concentration of ERM, SNCs aggregated to 437.3 nm in diameter in 30× ERM solution. Simultaneously, soluble silver chloro complexes (various combinations of [AgCl]0, [AgCl2]1−, [AgCl3]2−, and [AgCl4]3−) were calculated to have been formed. The patterns of the absorption spectra of SNCs and AgNO3 (a reference compound) differed markedly in ERM at different salinities, indicating that different soluble silver complexes were present in each solution. With increasing salinity, the chorion resistance decreased, and the salinity in the medaka eggs, as indicated by the osmotic pressure, increased. Simultaneously, uptake of SNCs or other silver complexes into the embryos also increased compared with that of AgNO3 in 20× and 30× ERM. In the presence of SNCs in 20× ERM, embryo hatching rate and full body lengths of post-hatch larvae were significantly lower than those with AgNO3. The toxic effects of SNCs on the hatching rate increased significantly in media of high salinity and were greater than those of AgNO3. SNCs and related silver chloro complexes exhibited higher bioavailability and medaka embryo toxicity in saline conditions than did AgNO3. SNCs pose greater ecological risks to fish embryos in high-salinity aquatic environments than in freshwater environments.

 Please wait while we load your content...
Please wait while we load your content...