Tin sulfide and selenide clusters soluble in organic solvents with the core structures of Sn4S6 and Sn4Se6†
Abstract
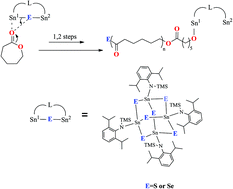
Reactions of LSnCl (1) (L = N(2,6-iPr2C6H3)(SiMe3)) with sulfur and selenium, respectively under mild conditions yielded two tin chalcogenide clusters. Surprisingly the tin atoms of the L4Sn4S6 (2) and L4Sn4Se6 (3) clusters are oxidized from Sn(II) of the precursor to Sn(IV) of the products under concomitant reduction of elemental sulfur and selenium to sulfide and selenide, respectively. The released chlorine radicals from the precursor LSnCl (1) react under oxidative addition with another LSnCl molecule to yield the side product LSnCl3 (4). The soluble nature of clusters 2 and 3 in organic solvents is a unique property of this class of compounds and makes them suitable for reactions in organic solvents. Compounds 2 and 3 were characterized by single crystal X-ray diffraction and multinuclear NMR investigations. Furthermore in ROP polymerization, the two products show high catalytic activity. For the first time a tin selenide compound functions in ROP catalysis.

 Please wait while we load your content...
Please wait while we load your content...