Luminescent europium and terbium complexes of dipyridoquinoxaline and dipyridophenazine ligands as photosensitizing antennae: structures and biological perspectives†
Abstract
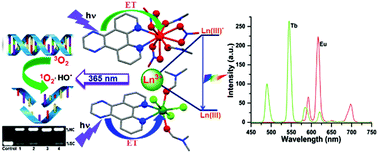
The europium(III) and terbium(III) complexes, namely [Eu(dpq)(DMF)2(NO3)3] (1), [Eu(dppz)2(NO3)3] (2), [Tb(dpq)(DMF)2Cl3] (3), and [Tb(dppz)(DMF)2Cl3] (4), where dipyrido[3,2-d:2′,3′-f]quinoxaline (dpq in 1 and 3), dipyrido[3,2-a:2′,3′-c]phenazine (dppz in 2 and 4) and N,N′-dimethylformamide (DMF) have been isolated, characterized from their physicochemical data, luminescence studies and their interaction with DNA, serum albumin protein and photo-induced DNA cleavage activity are studied. The X-ray crystal structures of complexes 1–4 show discrete mononuclear Ln3+-based structures. The Eu3+ in [Eu(dpq)(DMF)2(NO3)3] (1) and [Eu(dppz)2(NO3)3] (2) as [Eu(dppz)2(NO3)3]·dppz (2a) adopts a ten-coordinated bicapped dodecahedron structure with a bidentate N,N-donor dpq ligand, two DMF and three NO3− anions in 1 and two bidentate N,N-donor dppz ligands and three NO3− anions in 2. Complexes 3 and 4 show a seven-coordinated mono-capped octahedron structure where Tb3+ contains bidentate dpq/dppz ligands, two DMF and three Cl− anions. The complexes are highly luminescent in nature indicating efficient photo-excited energy transfer from the dpq/dppz antenna to Ln3+ to generate long-lived emissive excited states for characteristic f → f transitions. The time-resolved luminescence spectra of complexes 1–4 show typical narrow emission bands attributed to the 5D0 → 7FJ and 5D4 → 7FJ f–f transitions of Eu3+ and Tb3+ ions respectively. The number of inner-sphere water molecules (q) was determined from luminescence lifetime measurements in H2O and D2O confirming ligand-exchange reactions with water in solution. The complexes display significant binding propensity to the CT-DNA giving binding constant values in the range of 1.0 × 104–6.1 × 104 M−1 in the order 2, 4 (dppz) > 1, 3 (dpq). DNA binding data suggest DNA groove binding with the partial intercalation nature of the complexes. All the complexes also show binding propensity (KBSA ∼ 105 M−1) to the bovine serum albumin (BSA) protein. The intensity of the time-gated luminescence spectral bands enhances significantly with the increasing DNA concentration in aqueous buffer medium due to displacement of bound water upon interaction with DNA, thus reducing non-radiative quenching through the O–H oscillator. Complexes 1–4 efficiently cleave supercoiled (SC) ds-DNA to its nicked circular (NC) form on exposure to UV-A light of 365 nm via formation of singlet oxygen (1O2) and hydroxyl radicals (HO˙) as the reactive oxygen species at micromolar concentrations under physiological conditions.

 Please wait while we load your content...
Please wait while we load your content...