Novel La3Fe(MoO4)6 phase: magnetic properties and ethanol reactivity†
Abstract
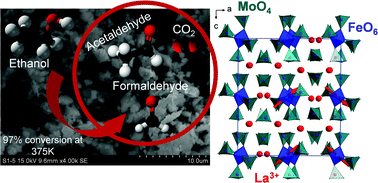
Single crystals of a new oxide, La3Fe(MoO4)6, were grown from fluxes of oxide precursors, and a polycrystalline sample was also prepared by a standard solid state reaction. La3Fe(MoO4)6 crystallizes in the orthorhombic space group Pbca with unit cell parameters a = 19.3164(11), b = 10.4143(5) and c = 22.0594(12) Å. This crystal structure exhibits a singular architectural type built on infinite chains of Fe(MoO4)4, each of them being surrounded by two isolated MoO4 tetrahedra and three isolated La3+ cations. Fe3+ ions in La3Fe(MoO4)6 are antiferromagnetically ordered below TN = 6.6 K in chains and between chains, as refined from neutron diffraction data. Further the redox stability of this compound – pure powder – was checked using temperature-programmed X-ray diffraction under a controlled atmosphere; under air, we observed a reversible phase transition above 523 K. The same phenomenon was observed under a reductive atmosphere, followed by a destruction of the as-formed phase above 923 K owing to iron III to II reduction. Reactivity of ethanol was then evaluated to get insights into the redox properties of the material under working conditions. After 4 hours of reaction at 648 K, the ethanol conversion was 97% with a selectivity to acetaldehyde of ∼60%, the other products being formaldehyde (∼10%) and CO2 (∼30%), underlining a better acetaldehyde selectivity than that of the La-free conventional Fe2(MoO4)3 catalytic formulation.

 Please wait while we load your content...
Please wait while we load your content...