Controlled in situ reaction for the assembly of Cu(ii) mixed-ligand coordination polymers: synthesis, structure, mechanistic insights, magnetism and catalysis†
Abstract
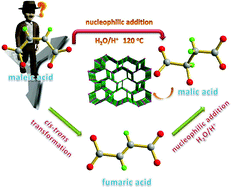
It has been a challenge to decipher the in situ ligand reaction mechanism in assembly processes, involving metals and ligands. The present study shows two crystalline mixed-ligand Cu(II) coordination polymers isolated by controlled in situ ligand reactions under the same hydrothermal conditions. Two closely related examples provide a precious chance to access the in-depth mechanistic issues surrounding in situ reactions. The solid structure of 1 demonstrated that maleic acid was completely transformed to malic acid, whereas when fumaric acid was used in the same reaction conditions, an incomplete transformation from fumaric acid to malic acid was observed in the solid structure of 2. Compound 1 exhibits a 3D 6-connected sni network based on a binuclear copper(II) secondary building unit, whereas the 3D network of 2 is classified to a (6,8)-connected topology network. The difference in steric hindrance between maleic acid and fumaric acid dictates the degree of the in situ nucleophilic addition reaction. The comparison of synthesis and final solid structures indicates that the one-step nucleophilic addition mechanism for in situ generated malic acid under the current reaction conditions of 1 is plausible. The magnetic sensitivity measurements of 1 demonstrated that overall antiferromagnetic coupling exists between Cu1 and Cu2 ions and between Cu3 and Cu4 ions. Furthermore, the obtained 1 can be an active catalyst for the solvent-free silylcyanation of aromatic aldehydes under mild conditions.


 Please wait while we load your content...
Please wait while we load your content...