A relationship between structural and electronic order–disorder effects and optical properties in crystalline TiO2 nanomaterials†
Abstract
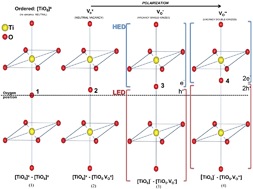
The focus of this paper is on the analysis of the structural and electronic order–disorder effects at long, medium and short ranges of titanium dioxide (TiO2) nanoparticles synthesized by the sol–gel process followed by the microwave-assisted solvothermal (MAS) method at low temperatures and short reaction times. X-ray diffraction (XRD), Rietveld refinement, micro-Raman (MR) spectroscopy, transmission electron microscopy (TEM) and X-ray spectroscopy (EDX) were used to characterize the TiO2 nanoparticles. Optical properties were investigated by ultraviolet-visible (UV-vis) and photoluminescence (PL) measurements performed at room temperature. XRD and Rietveld refinement confirmed the presence of the anatase and brookite phases; nonetheless anatase is the major phase. The X-ray photoelectron spectroscopy (XPS) analysis revealed the presence of only Ti4+ but the nonstoichiometry revealed that TiO2 NPs contain defects assigned to oxygen vacancies that lead to structural and electronic order–disorder effects observed by band gap narrowing and PL wide band emission. These intermediary energy levels (shallow and deep levels) created within the band gap act as acceptors/donors of electrons and recombination centers. The oxygen vacancies (VOx, VO˙ and VO˙˙) responsible by degree of structural order–disorder are related to distortions (tilts) on the [TiO6] octahedron and changes in the bond lengths and bond angles between oxygen and titanium atoms that gave rise to new species of cluster makers such as [TiO6]′, [TiO5·VOx], [TiO5·VO˙] and [TiO5·VO˙˙]. This structural transformation is consistent with a redistribution of electron density from highly ordered [TiO6]x clusters which form distorted [TiO6]′ as well as complex [TiO5·VOx], [TiO5·VO˙] and [TiO5·VO˙˙] clusters assigned to oxygen vacancies which were understood as displacements in the oxygen atoms’ position in the bond lengths (Ti–O).

 Please wait while we load your content...
Please wait while we load your content...