Amorphous Co3O4 modified CdS nanorods with enhanced visible-light photocatalytic H2-production activity
Abstract
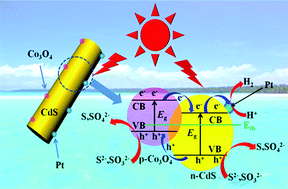
In this work, amorphous Co3O4 modified CdS nanorods were synthesized by a two-step solvothermal/hydrothermal method, and characterized by transmission electron microscopy (TEM), X-ray diffraction (XRD), high-resolution transmission electron microscopy, UV-visible spectroscopy, nitrogen absorption and X-ray photoelectron spectroscopy. The photocatalytic performance of the as-synthesized Co3O4–CdS nanorods was evaluated through H2 generation from an aqueous solution containing sulfide and sulfite under visible light (λ ≥ 420 nm). The results showed that the photocatalytic activity of CdS nanorods for H2 evolution could be significantly enhanced by loading the amorphous Co3O4. The optimal Co3O4 loading was found to be approximately 3.0 mol%. The as-prepared CdS nanorods with 3 mol% Co3O4 exhibited the highest photocatalytic activity for H2 evolution under visible light irradiation, 236 μmol g−1 h−1, which is 33-fold higher than that of the pristine CdS nanorods. Furthermore, the co-loading of 1 wt% Pt can lead to another three times enhancement in the photocatalytic H2-production activity. The mechanism for the enhanced H2-production performance of Co3O4–CdS nanorods was discussed. The excellent performance of Co3O4–CdS nanorods is mainly ascribed to the loading of amorphous Co3O4 onto the surface of CdS nanorods, which could promote the separation of electron–hole pairs and enhance the stability of CdS nanorods due to the formation of p–n heterojunctions between the Co3O4 and CdS nanorods, thus leading to an enhanced activity for H2 generation. This work demonstrated that the loading of amorphous Co3O4 is a facile strategy to enhance the photocatalytic activity of CdS nanorods, which may provide some potential opportunities for designing other composite photocatalysts for water splitting.

 Please wait while we load your content...
Please wait while we load your content...