A biodiesel additive: etherification of 5-hydroxymethylfurfural with isobutene to tert-butoxymethylfurfural
Abstract
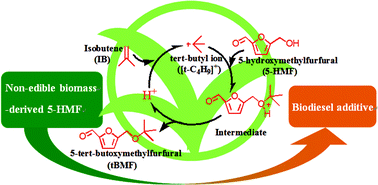
Biodiesel is a sustainable and environmentally compatible diesel fuel substitute that presents certain limitations, particularly low-temperature flow properties, which are attributable to its long fatty chain structure. Branched alkyl derivates such as tertiary butyl ethers are efficient biodiesel additives for improved cold flow behaviour of biodiesel. In this study, tert-butoxymethylfurfural (tBMF) is synthesised as a biodiesel additive through etherification of 5-hydroxymethylfurfural (5-HMF), a non-edible biomass-derived building block, with isobutene (IB) on a series of acid zeolites and liquid acids. The catalytic activity and selectivity of a zeolite catalyst involve not only optimisation of the number and strength of its acidic sites and adsorption properties but also promotion of its internal pore space. Efficient solvent systems did not include extremely weakly polar or strongly polar solvents but included moderately polar solvents, such as glycol dimethyl ether which presents suitable hydrophilic/hydrophobic properties. The HY zeolite with a SiO2/Al2O3 mole ratio of 12 in combination with glycol dimethyl ether solvent exhibited an excellent tBMF selectivity of 94 mol% with 59 mol% 5-HMF conversion after 3 h of reaction at 60 °C. Side reactions of 5-HMF dimerisation and the IB oligomerisation were minimal under mild reaction conditions. The activity of the deactivated catalyst was fully recovered by calcination in air, and a detailed deactivation mechanism was proposed.

 Please wait while we load your content...
Please wait while we load your content...