Core-structure-inspired asymmetric addition reactions: enantioselective synthesis of dihydrobenzoxazinone- and dihydroquinazolinone-based anti-HIV agents
Abstract
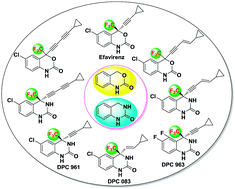
Dihydrobenzoxazinones and dihydroquinazolinones are the core units present in many anti-HIV agents, such as Efavirenz, DPC 961, DPC 963, and DPC 083. All these molecules contain a trifluoromethyl moiety at the quaternary stereogenic carbon center with S configuration. The enantioselective addition of carbon nucleophiles to ketones or cyclic ketimines could serve as a key step to access these molecules. This tutorial review provides an overview of significant advances in the synthesis of dihydrobenzoxazinone- and dihydroquinazolinone-based anti-HIV agents and relative analogues, with an emphasis on asymmetric addition reactions for the establishment of the CF3-containing quaternary carbon centers.

 Please wait while we load your content...
Please wait while we load your content...