Gas-phase chemistry of technetium carbonyl complexes
Abstract
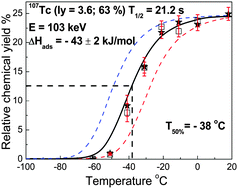
Gas-phase chemical behaviors of short-lived technetium carbonyl complexes were studied using a low temperature isothermal chromatograph (IC) coupled with a 252Cf spontaneous fission (SF) source. Fission products recoiled from the 252Cf SF source were thermalized in a mixed gas containing CO, and then technetium carbonyl complexes were formed from reactions between CO gas and various technetium isotopes. A gas-jet system was employed to transport the volatile carbonyl complexes from a recoil chamber to the IC. Short IC columns made of Fluorinated Ethylene Propylene (FEP) Teflon and quartz were used to obtain chemical information about the technetium carbonyl complexes. The results for the 104Tc–106Tc carbonyl complexes were found to be strongly influenced by the precursors, and showed the chemical behaviors of 104Mo–106Mo carbonyl complexes, respectively. However, 107Tc and 108Tc could represent the chemical information of the element technetium due to their high independent yields and the very short half-lives of their precursors 107Mo and 108Mo. An adsorption enthalpy of about ΔHads = −43 kJ mol−1 was determined for the Tc carbonyl complexes on both the Teflon and quartz surfaces by fitting the breakthrough curves of the 107Tc and 108Tc carbonyl complexes with a Monte Carlo simulation program. Chemical yields of around 25% were measured for the Tc carbonyl complexes relative to the transport yields obtained with the gas-jet transport of KCl aerosol particles with Ar carrier gas. Furthermore, the influence of a small amount of O2 gas on the yields of the Mo and Tc carbonyl complexes was studied.

 Please wait while we load your content...
Please wait while we load your content...