Solvent-dependent structure of molecular iodine probed by picosecond X-ray solution scattering†
Abstract
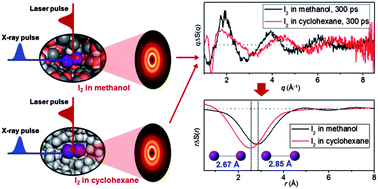
The effect of solute–solvent interaction on molecular structure and reaction dynamics has been a target of intense studies in solution-phase chemistry, but it is often challenging to characterize the subtle effect of solute–solvent interaction even for the simplest diatomic molecules. Since the I2 molecule has only one structural parameter and exhibits solvatochromism, it is a good model system for investigating the solvent dependence of the solute structure. By using X-rays as a probe, time-resolved X-ray liquidography (TRXL) can directly elucidate the structures of reacting molecules in solution and can thus determine the solvent-dependent structural change with atomic resolution. Here, by applying TRXL, we characterized the molecular structure of I2 in methanol and cyclohexane with sub-angstrom accuracy. Specifically, we found that the I–I bond length of I2 is longer in the polar solvent (methanol) by ∼0.2 Å than in nonpolar solvents (cyclohexane and CCl4). Density functional theory (DFT) using 22 explicit methanol molecules well reproduces the longer I–I bond of molecular iodine in methanol and reveals that the larger bond length originates from partial negative charge filled in an antibonding σ* orbital through solvent-to-solute charge transfer.


 Please wait while we load your content...
Please wait while we load your content...