Competitive excited-state single or double proton transfer mechanisms for bis-2,5-(2-benzoxazolyl)-hydroquinone and its derivatives†
Abstract
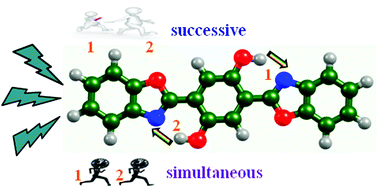
The excited state intramolecular proton transfer (ESIPT) mechanisms of 2-(2-hydroxyphenyl)benzoxazole (HBO), bis-2,5-(2-benzoxazolyl)-hydroquinone (BBHQ) and 2,5-bis(5′-tert-butyl-benzoxazol-2′-yl)hydroquinone (DHBO) have been investigated using time-dependent density functional theory (TDDFT). The calculated vertical excitation energies based on the TDDFT method reproduced the experimental absorption and emission spectra well. Three kinds of stable structures were found on the S1 state potential energy surface (PES). A new ESIPT mechanism that differs from the one proposed previously (Mordzinski et al., Chem. Phys. Lett., 1983, 101, 291. and Lim et al., J. Am. Chem. Soc., 2006, 128, 14542.) is proposed. The new mechanism includes the possibility of simultaneous double proton transfer, or successive single transfers, in addition to the accepted single proton transfer mechanism. Hydrogen bond strengthening in the excited state was based on primary bond lengths, angles, IR vibrational spectra and hydrogen bond energy. Intramolecular charge transfer based on the frontier molecular orbitals (MOs) also supports the proposed mechanism of the ESIPT reaction. To further elucidate the proposed mechanism, reduced dimensionality PESs of the S0 and S1 states were constructed by keeping the O–H distance fixed at a series of values. The potential barrier heights among the local minima on the S1 surface imply competitive single and double proton transfer branches in the mechanism. Based on the new ESIPT mechanism, the observed fluorescence quenching can be satisfactorily explained.

 Please wait while we load your content...
Please wait while we load your content...