Oxygen nonstoichiometry and thermodynamic characterization of Zr doped ceria in the 1573–1773 K temperature range†
Abstract
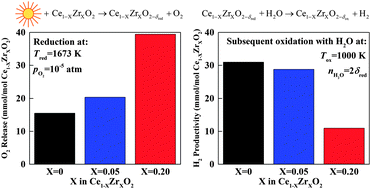
This work encompasses the thermodynamic characterization and critical evaluation of Zr4+ doped ceria, a promising redox material for the two-step solar thermochemical splitting of H2O and CO2 to H2 and CO. As a case study, we experimentally examine 5 mol% Zr4+ doped ceria and present oxygen nonstoichiometry measurements at elevated temperatures ranging from 1573 K to 1773 K and oxygen partial pressures ranging from 4.50 × 10−3 atm to 2.3 × 10−4 atm, yielding higher reduction extents compared to those of pure ceria under all conditions investigated, especially at the lower temperature range and at higher pO2. In contrast to pure ceria, a simple ideal solution model accounting for the formation of isolated oxygen vacancies and localized electrons accurately describes the defect chemistry. Thermodynamic properties are determined, namely: partial molar enthalpy, entropy, and Gibbs free energy. In general, partial molar enthalpy and entropy values of Zr4+ doped ceria are lower. The equilibrium hydrogen yields are subsequently extracted as a function of the redox conditions for dopant concentrations as high as 20%. Although reduction extents increase greatly with dopant concentration, the oxidation of Zr4+ doped ceria is thermodynamically less favorable compared to pure ceria. This leads to substantially larger temperature swings between reduction and oxidation steps, ultimately resulting in lower theoretical solar energy conversion efficiencies compared to ceria under most conditions. In effect, these results point to the importance of considering oxidation thermodynamics in addition to reduction when screening potential redox materials.

 Please wait while we load your content...
Please wait while we load your content...