Genetics of superionic conductivity in lithium lanthanum titanates†
Abstract
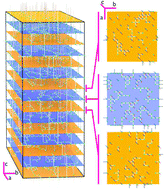
The self-diffusion of ions is a fundamental mass transport process in solids and has a profound impact on the performance of electrochemical devices such as the solid oxide fuel cell, batteries and electrolysers. The perovskite system lithium lanthanum titanate, La2/3−xLi3xTiO3 (LLTO) has been the subject of much academic interest as it displays very high lattice conductivity for a solid state Li conductor; making it a material of great technological interest for deployment in safe durable mobile power applications. However, so far, a clear picture of the structural features that lead to efficient ion diffusion pathways in LLTO, has not been fully developed. In this work we show that a genetic algorithm in conjunction with molecular dynamics can be employed to elucidate diffusion mechanisms in systems such as LLTO. Based on our simulations we provide evidence that there is a three-dimensional percolated network of Li diffusion pathways. The present approach not only reproduces experimental ionic conductivity results but the method also promises straightforward investigation and optimisation of the properties relating to superionic conductivity in materials such as LLTO. Furthermore, this method could be used to provide insights into related materials with structural disorder.

 Please wait while we load your content...
Please wait while we load your content...