Sequential single-crystal-to-single-crystal transformations promoted by gradual thermal dehydration in a porous metavanadate hybrid†
Abstract
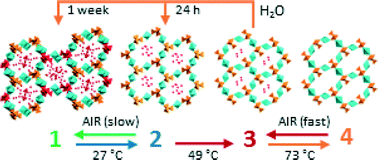
TheE porous hybrid metavanadate [{Cu(cyclam)}(VO3)2]·5H2O (1) (cyclam = 1,4,8,11-tetraazacyclotetradecane) undergoes thermally-triggered sequential single-crystal-to-single-crystal (SCSC) transformations upon gradual dehydration to produce three new porous crystalline phases, namely [{Cu(cyclam)}(VO3)2]·3H2O (2), [{Cu(cyclam)}(VO3)2]·1.3H2O (3) and [{Cu(cyclam)}(VO3)2] (4). Compound 1 has a three-dimensional structure formed by metavanadate chains linked by {Cu(cyclam)} moieties in a hybrid open framework with two different types of hexagonal channels where water molecules of hydration are hosted. The SCSC transformations cause a rearrangement o/f the metavanadate chains in such a way that they contract when going from 1 to 2 and stretch back when 2 transforms into 3. The size of the channels is also modified as the hybrid dehydrates. The transition from 2 to 3 leads to the cleavage of a Cu–O bond and consequent coordination of a {Cu(cyclam)} moiety to a different {VO4} unit, which drastically decreases the size of the channel in the process. In contrast, total dehydration of 3 enlarges the channel in the anhydrous phase 4 due to the migration of another Cu atom. The reversibility of such transformations has been monitored by a combination of thermogravimetric and powder X-ray diffraction analyses. While 1 and 3 are stable in open-air conditions, 2 transforms back into 1 upon exposure to air for three weeks. The anhydrous phase 4 rapidly rehydrates into 3 when in contact with ambient moisture. Furthermore, 3 can also be transformed into the parent hybrid 1 when soaked in water for seven days. Therefore, all crystal transitions described herein are fully reversible by applying the appropriate conditions.
- This article is part of the themed collection: Single-Crystal-to-Single-Crystal Transformations

 Please wait while we load your content...
Please wait while we load your content...