A new colorimetric protocol for selective detection of phosphate based on the inhibition of peroxidase-like activity of magnetite nanoparticles†
Abstract
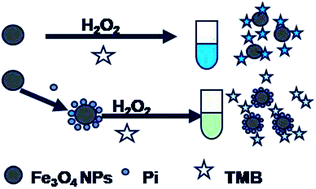
A simple colorimetric assay for phosphate ion (Pi) has been established based on analyte-induced inhibition of the magnetite nanoparticles (MNPs)-catalyzed oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB) in the presence of H2O2. The Fe3O4 MNPs can catalyze the H2O2-mediated oxidation of TMB and yields a blue oxidized product, which exhibits a maximum absorption at 652 nm. Pi could be absorbed on the surface of the Fe3O4 MNPs through coordinating with Fe3+, inducing a reduced colorimetric signal. The colorimetric signal change (ΔA652) in this process was proportional to the concentration of Pi, ranging from 0.2 μM to 200 μM. The limit of detection (S/N = 3) was as low as 0.11 μM. The as-proposed Fe3O4 MNPs–TMB–H2O2 probe exhibited a high selectivity toward Pi over other relevant ions that commonly exist in water and has been applied to Pi detection in drinking water, ground water and lake water samples with satisfactory results.

 Please wait while we load your content...
Please wait while we load your content...