A step towards mobile arsenic measurement for surface waters
Abstract
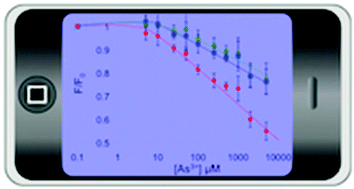
Surface modified quantum dots (QDs) are studied using a bio-inspired cysteine rich ligand (glutathione, GSH) and their quenching response and selectivity to arsenic examined. As predicted from As3+ binding with highly crosslinked phytochelatin-(PCn)-like molecules, better arsenic selectivity is obtained for a thicker more 3-dimensional GSH surface layer, with exposed sulfhydryl groups. A detection limit of at least 10 μM can be achieved using CdSe/ZnS core–shell QDs capped with this GSH structure. The system is also demonstrated using a mobile phone camera to record the measurement, producing a detection limit of 5 μM. However, copper remains the main interferent of concern. Water-soluble CdTe QDs show little sensitivity to As3+ even with a GSH surface, but they remain sensitive to Cu2+, allowing a copper baseline to be established from the CdTe measurement. Despite anticipating that spectrally non overlapping fluorescence would be required from the two types of QDs to achieve this, a method is demonstrated using RGB channels from a mobile phone and processing the raw data for CdTe QDs, with an emission wavelength of 600 nm, and CdSe/ZnS QDs, with emission maximum of 630 nm. It is shown that As3+ measurement remains feasible at the WHO guideline value of 10 μg L−1 up to a copper concentration of around 0.3 μM Cu2+, which corresponds to the highest recorded level in a selection of large rivers world-wide.
- This article is part of the themed collection: Analytical Sciences in the UK

 Please wait while we load your content...
Please wait while we load your content...