A simple and highly sensitive fluorescence assay for microRNAs†
Abstract
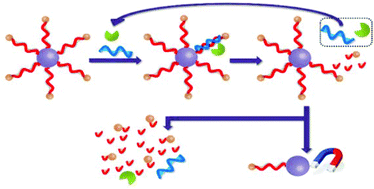
Herein, we have reported a simple and highly sensitive fluorescence assay for the detection of microRNAs (miRNAs). The assay uses a duplex-specific nuclease (DSN) to amplify the fluorescence signal and magnetic beads (MBs) to completely remove the unreacted DNA detection probes. Briefly, fluorescein-capped DNA detection probes were first conjugated to the MBs. The use of the MBs produced a very low background signal since all the unreacted DNA probes can be conveniently removed from the solution by using a permanent magnet. During the assaying process, target miRNA strands hybridized with the DNA capture probes to form miRNA–DNA heteroduplexes. The DSN then selectively cleaved the DNA probes in the miRNA–DNA duplexes and release the target miRNA strands back into the solution, thereby establishing a target recycling amplification mechanism – a cumulative signal amplification process. A much-amplified fluorescence signal was obtained in the presence of traces of the target miRNA. In addition, a negligible background signal was conveniently attained by the complete removal of the unreacted DNA detection probes so that minute change in the fluorescence signal can be unambiguously detected. The negligible background signal in association with the accumulative signal amplification significantly lowered the detection limit and broadened the dynamic range of the assay. Moreover, the high specificity of the DSN to perfectly matched duplexes endowed this assay with good selectivity when analyzing target miRNAs with high sequence similarities. Successful attempts were made in applying the proposed assay to detect let-7a in total RNA extracted from cultured cells.

 Please wait while we load your content...
Please wait while we load your content...